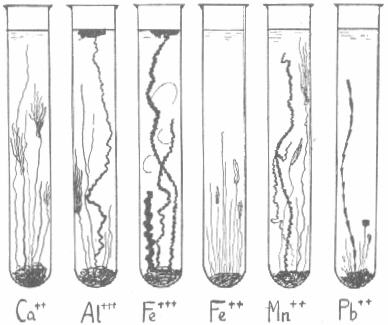


Each metallic salt produces within a few minutes its characteristic structure as a result of osmotic pressure and the breaking of the insoluble membrane of metal silicate. Previous addition of small amounts of sodium carbonate or phosphate to the sodium silicate effects a change in structure.
Most active are the salts of Fe, Ni, Co, Mn, Al, Mg, Ca, Ba. Least active are Cu, Pb, U.